DynaForce® Staple Compression Plates™
Give Fusions the G.A.S.!

Our proprietary Staple Compression Plates™ (SCPs) are the only plates which utilize a staple instead of a lag screw to provide compression. This gives three important advantages:
Gap Recovery
Compressing the fusing joint if there is bone resorption
Apposition
Provides full bone surface contact
Speed
Reduces OR time by up to an hour
SCP gap recovery, apposition and speed (GAS) all work to
give faster fusions and rapid recovery.
DynaForce® Staple Compression Plates
Gap Recovery Simulation
Watch a simulation of DynaForce® SCP’s gap recovery versus conventional plate and cross screw construct.
Complete Bone Apposition
One of the least discussed features of the SCP™ system is the staple does not violate the surface area of the fusion site like a lag screw. This bone preservation may have a substantial positive impact on bone healing.
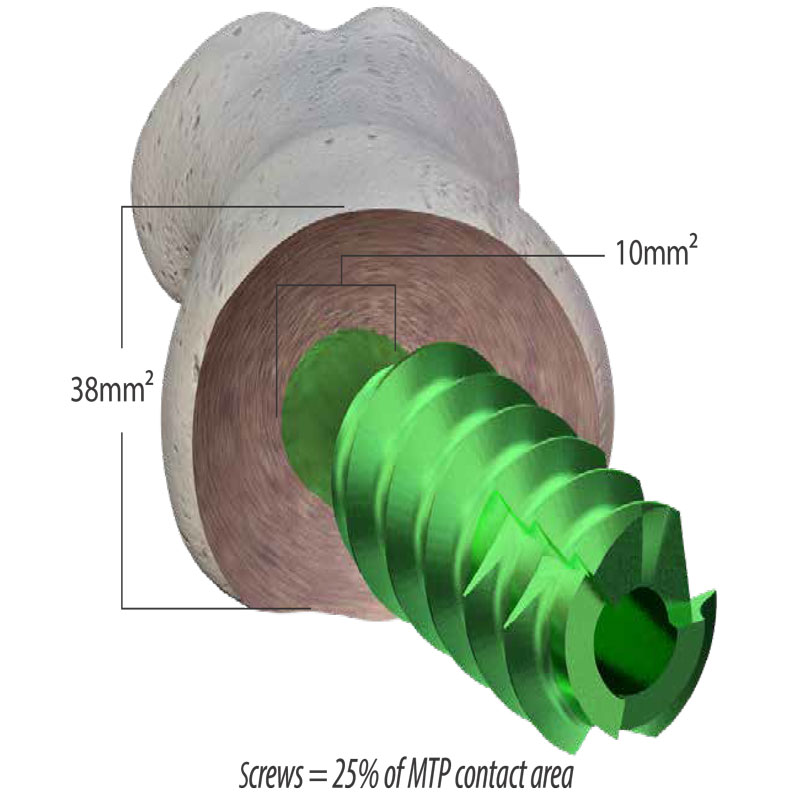

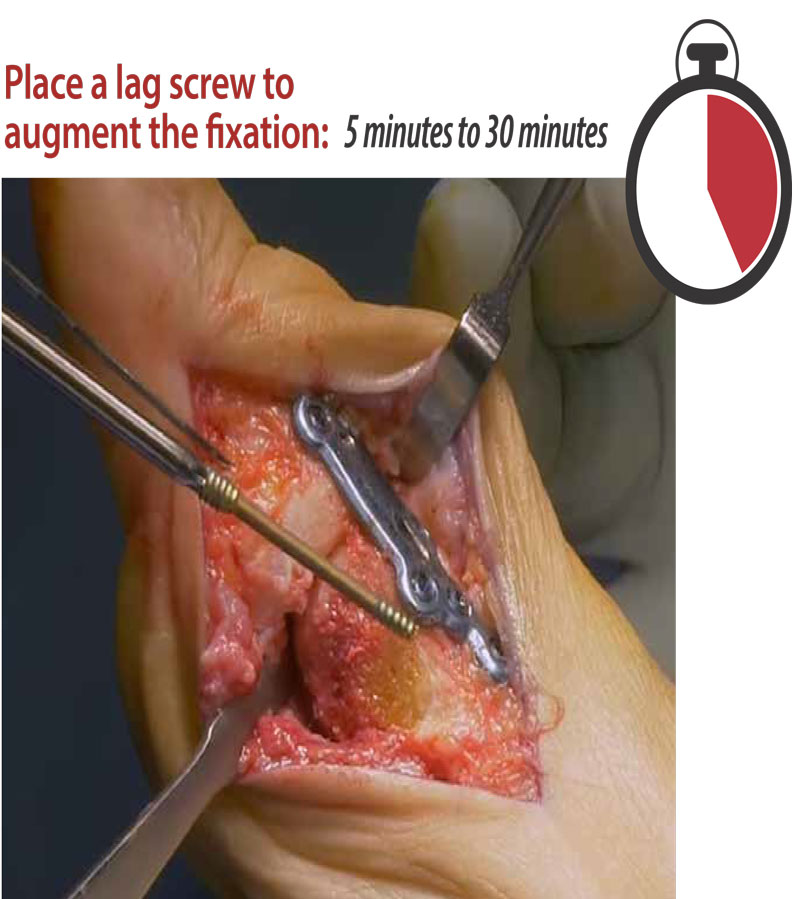
Speed
Traditional fusion plates require a fixation screw (lag screw) to be driven through the joint for added fixation and compression. However, placing a lag screw is time-consuming due to trajectory and anatomic issues. The staple of the DynaForce® delivers compression much more efficiently.
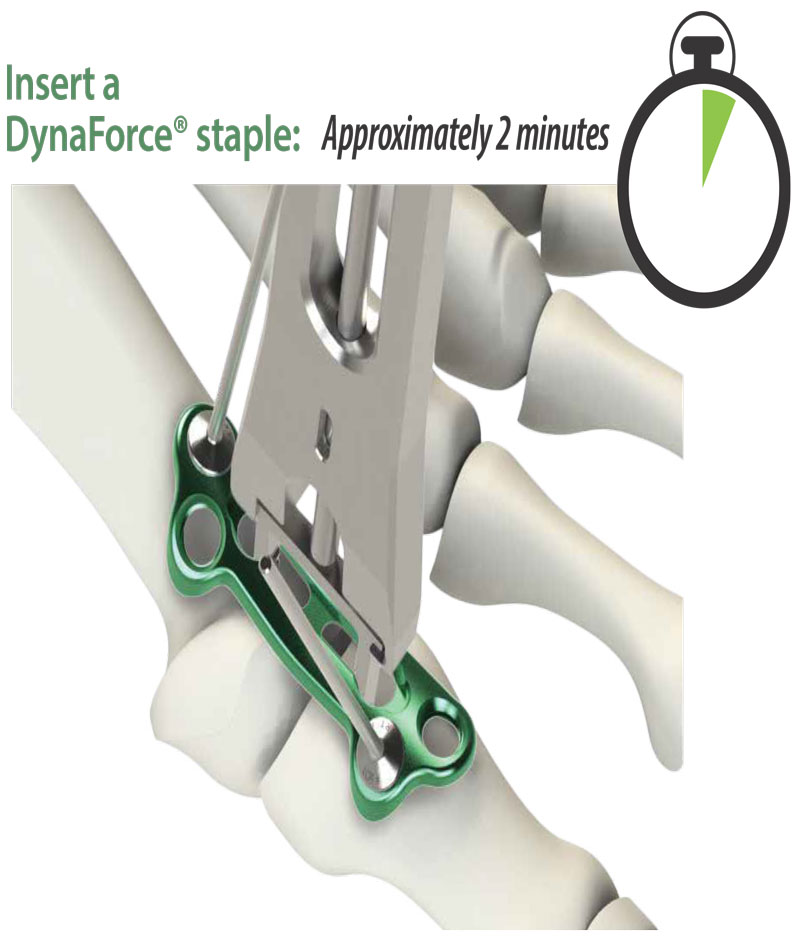
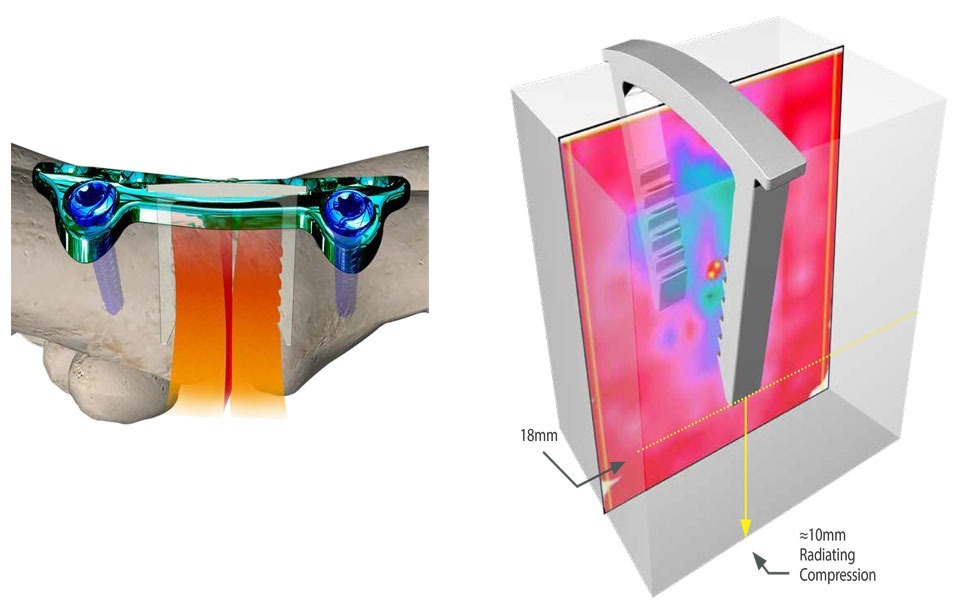
Gap Recovery Through Deep Staple Compression
The DynaFORCE® staple system boasts 27lbs. of strong compression.
Additionally, independent testing has demonstrated the compression of the staple radiates deep into the bone to reduce gaps in the fusion. This allows better bone contact during the healing process to create a stronger fusion*.
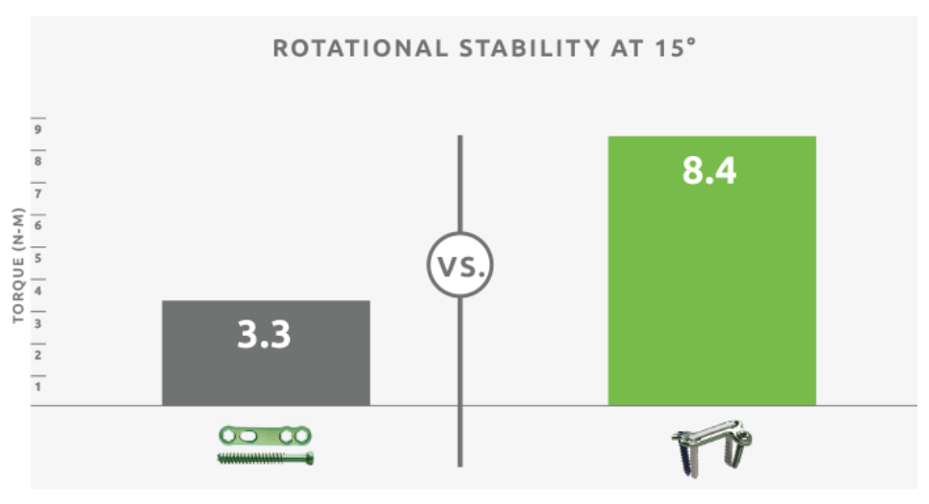
Rotational Stability
The DynaForce® Staple Compression Plate System has 2.5 times more rotational stability than a
traditional plate and lag screw.
Family of Products

VERO® Plating System
with DynaForce® SCP Technology

Versatile Design
The plates are anatomically designed with three options (proximal, distal or tri-joint) to address deformities in the medial column.
Accommodates Multiple Staples
Built to accommodate multiple HiMax® staples
(15mm or 18mm)
Formability of Wings
Unique plate design allows conformance to anatomy
First TMT Fusion with
DynaForce® SCP Technology
Optimized Anatomic Design
The DynaForce® Lapidus implant system has a low profile anatomic plate design and is available in both a left and right
configuration with an 18mm staple option.

First Metatarsophalangeal Joint Fusion
with DynaForce® SCP Technology
Optimized Plate Designs Specifically for MTP
The DynaForce® MTP plates are low profile and designed to match the anatomy of the patient. We offer multiple plate sizes and 0, 5 and 10 degree dorsiflexion.
Applicable for Revision Indications
The simplicity, stability and continuous compression afforded by the DynaForce® MTP system has been found particularly advantageous for revising failed reconstructive procedures. Our system features Extended plate sizes and longer staples which are well-suited for these indications.

Z Plates for Talonavicular™
or Lapidus Fusion with DynaForce® SCP Technology
Anatomic Design
The talonavicular (TN) Fusion implant system has a low profile anatomic plate
design with both 15mm and 18mm staple options.
Our Z plates have a patented geometry that delivers much more
stability than a standard straight or anatomic plate. These plates are generally utilized for TN fusions or Lapidus procedures due their ability to provide multi-planar, rigid stability.

Tarsometatarsal Fusion
with DynaForce® SCP Technology
Versatile Low Profile Design
The DynaForce® TMT 4-hole and 5-hole plates both have a low profile anatomic plate design. This system provides versatile offerings with 15mm and 18mm staple options in both plates

The MotoBAND™ CP Implant System is indicated for stabilization and fixation of fresh fractures, revision procedures, joint fusion and reconstruction of small bones of the hand, feet, wrist, ankles, fingers and toes. When used for these indications, the MotoBAND™ CP Implant System with the exception of the 2-hole plate may be used with the MotoCLIP™ Implant System.
There are potential risks associated with the use of these devices, some of which include: allergic reaction to the implant material, fracture of the implant, soft-tissue complication (e.g., infection at the implant site, prolonged healing), and revision surgery. Refer to IFU for all contraindications, warnings, and risks.
Contact Information
Use the contact form to let us know how we can help you!
For more information on miniBunion® go to www.miniBunion.com
For more information on dynaBunion® go to www.4Dbunion.com
Click here to find a foot doctor.
HEADQUARTERS
6423 Shelby View Dr. Suite 101,
Memphis, TN 38134
INFORMATION
Phone: (901) 221-8406
